 
This makes it less able to gain an extra electron. The outer electrons are further away from the nucleus with each additional electron shell making the force of attraction weaker.The reactivity decreases as you go down the group because: In a chemical reaction an extra electron is added to the outer shell. Halogens are toxic to bacteria and are used in disinfectants. Halogens all produce acids when combined with Hydrogen. Halogens all react strongly as bleaching agents. Group 7: The Halogens Chemical Properties The reactivity of Halogens decreases as you go down the Periodic Table. Alkali Metals all appear shiny (before they oxidise). 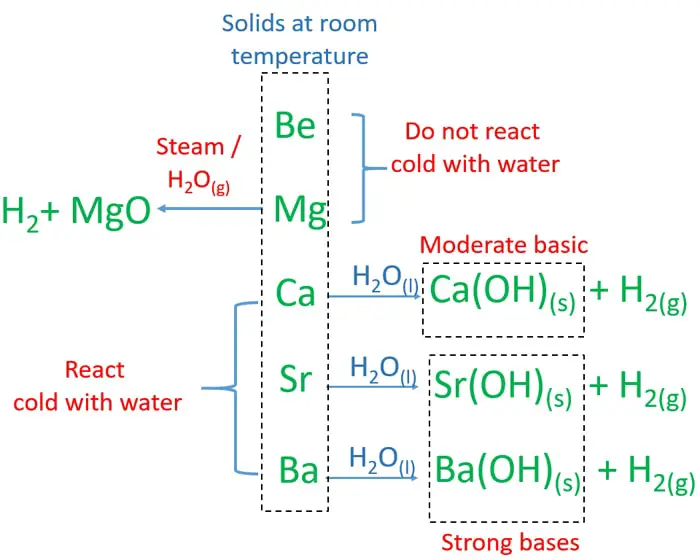
Alkali Metals are soft and can be easily cut. Alkali Metals are all solid at room temperature but have a low melting point compared to other metals. Physical Properties Alkali Metals have a low density compared to other metals. Even though the charge of the nucleus increases the outer electron is shielded from most of the positive charge of the nucleus by electrons in the inner shells.This makes it easier for an atom to lose it's outer electron. The outer electron is further away from the nucleus with each additional electron shell making the force of attraction weaker.The reactivity increases as you go down the group because: In a chemical reaction the electron in the outer shell is lost. It was later discovered that elements have similar chemical properties when they have the same number of electrons in their Outer Shells. The elements were oringally arranged groups of similar chemical properties. However, the property changes gradually as you move down the group.Ī Group is a column on the Periodic Table with elements with the same number of electrons on the Outer Shell.Ībout Groups Helium, in Group 0 is an exception to this rule as it has 2 electrons in its Outer Shell. The physical properties of elements within a group are similar. Group 0: The Noble Gases are all inert (unreactive).The reactivity decreases as you go down the group. Group 7: The Halogens all act as bleaching agents and kill bacteria.The reactivity increases as you go down the group. Group 2: The Alkali Earth Metals all react strongly with steam and acids.Group 1: The Alkali Metals all react strongly with water.However, the reactivity within a group changes as you move up or down the periods. The chemical properties of elements within a group are similar. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
